What Happens During a Clinical Trial? A Step-by-Step Guide for Participants
One of the most common questions people ask before joining a clinical trial is simple: What actually happens if I participate?
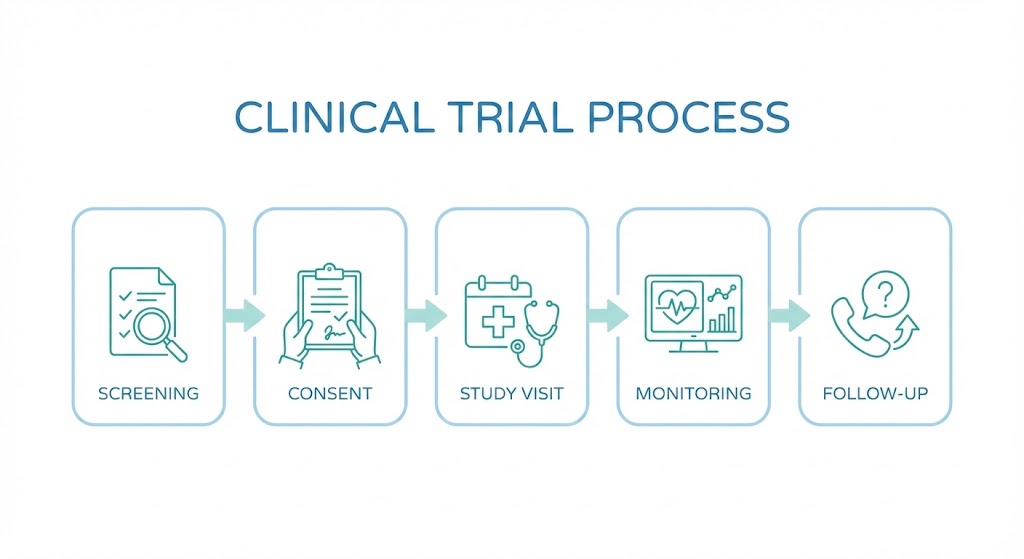
Understanding the process ahead of time can help reduce uncertainty and make it easier to decide whether a study is right for you. While every clinical trial is different, most follow a similar structure. This guide walks through the typical stages of a clinical trial from a participant’s perspective.
Step 1: Pre-Screening and Initial Contact
The first step usually begins when you express interest in a study. This may happen through an online form, phone call, or referral.
During pre-screening, the research team asks basic questions about:
-
Age
-
General health
-
Medical history
-
Current medications
-
Symptoms or conditions relevant to the study
This step helps determine whether the study may be a potential fit before moving forward.
Step 2: Eligibility Screening
If the pre-screening looks promising, you may be invited for a more detailed screening visit. This stage confirms whether you meet the study’s eligibility criteria.
Screening may include:
-
Review of medical history
-
Physical examination
-
Vital signs
-
Laboratory tests or questionnaires
Not everyone who is screened will qualify, and that’s normal. Eligibility criteria exist to protect participant safety and ensure reliable research results.
Step 3: Informed Consent
Before any study-related procedures begin, you will go through the informed consent process.
The research team will explain:
-
The purpose of the study
-
What participation involves
-
How long the study lasts
-
Possible risks and potential benefits
-
Your rights as a participant
You are encouraged to take your time, ask questions, and discuss participation with family or healthcare providers if needed. Participation only moves forward once you voluntarily agree.
Step 4: Study Visits and Participation
Once enrolled, you’ll begin the active phase of the clinical trial. This may involve one or multiple visits depending on the study design.
Study visits can include:
-
Health assessments
-
Study-related treatments or interventions
-
Questionnaires or interviews
-
Routine monitoring and follow-ups
The research team provides clear instructions and support throughout the study to ensure you know what to expect at each visit.
Step 5: Ongoing Monitoring and Communication
Participant safety and well-being are closely monitored throughout the study. You’ll have regular check-ins with the research team, and any changes in your health can be discussed at any time.
If new information becomes available during the study, the team will communicate this with you promptly. You always have the right to ask questions or raise concerns.
Step 6: Study Completion and Follow-Up
After completing the study, there may be a final visit or follow-up period. This helps researchers gather complete data and ensure your well-being after participation ends.
Some studies include:
-
Final health assessments
-
Post-study check-ins
-
Long-term follow-up contact
Once the study concludes, your participation officially ends unless you choose to join a related follow-up study.
Your Rights Throughout the Process
At every stage, participation remains voluntary. You can choose to withdraw from the study at any time without penalty or impact on your regular medical care.
The research team is there to support you, answer questions, and ensure you feel informed and respected throughout the process.
Final Thoughts
Knowing what happens during a clinical trial can make participation feel less uncertain and more approachable. Clinical research is structured, carefully monitored, and designed with participant safety at its core.
If you’re considering a study, understanding the journey ahead is an important step toward making an informed choice.
